Silicon Electron Configuration Orbital Diagram For Silicon (Si)
Symbol and electron diagram for silicon Royalty Free Vector
Silicon Bohr Model: Diagram, Steps To Draw Silicon is a group 14 element and is denoted by the symbol Si. It has a high affinity towards oxygen due to which it rarely occurs in its pure form in nature. It is a metalloid and a non-metal. It is the second most abundant mineral found on the earth's surface.
Bohr Model Silicon Atom Electron Structure Stock Vector (Royalty Free
Bohr Diagram: The First Element In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we'll show a sample Bohr diagram for hydrogen. H —Hydrogen 1 proton 1 electron 0 neutrons
PPT Chapter 5 The Periodic Law PowerPoint Presentation, free
On the far left of Figure 3.6.1 3.6. 1 are the highest energy electromagnetic waves. These are called gamma rays and can be quite dangerous, in large numbers, to living systems. The next lower energy form of electromagnetic waves are called x-rays. Most of you are familiar with the penetration abilities of these waves.
Silicon Bohr Model Diagram, Steps To Draw Techiescientist
In this video we'll look at the atomic structure and Bohr model for the Silicon atom (Si). We'll use a Bohr diagram to visually represent where the electrons.
Silicon Si (Element 14) of Periodic Table Elements FlashCards
Manish Bhardwaj. 5.6: Bohr Diagrams of Atoms and Ions is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells,..
Silicon Electron Configuration Orbital Diagram For Silicon (Si)
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,

WebElements Periodic Table » Silicon » properties of free atoms
The Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν). The orbits in which the electron may travel are shown as grey circles; their.
Silicon Bohr Diagram My XXX Hot Girl
Bohr Model of Silicon - Louisiana Tech University
Silicon Atom Bohr Model Cartoon Style Stock Vector (Royalty Free
Add one at a time -starting on the right side and going counter clock-wise. Check your work. You should have 6 total electrons for Carbon. Only two electrons can fit in the 1st shell. The 2nd shell can hold up to 8 electrons. The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons.
Bohr Model For Silicon
0:00 / 2:13 How to Draw the Bohr-Rutherford Diagram of Silicon chemistNATE 258K subscribers Subscribe Subscribed 245 22K views 4 years ago Silicon has 2 electrons in its first shell, 8 in its.
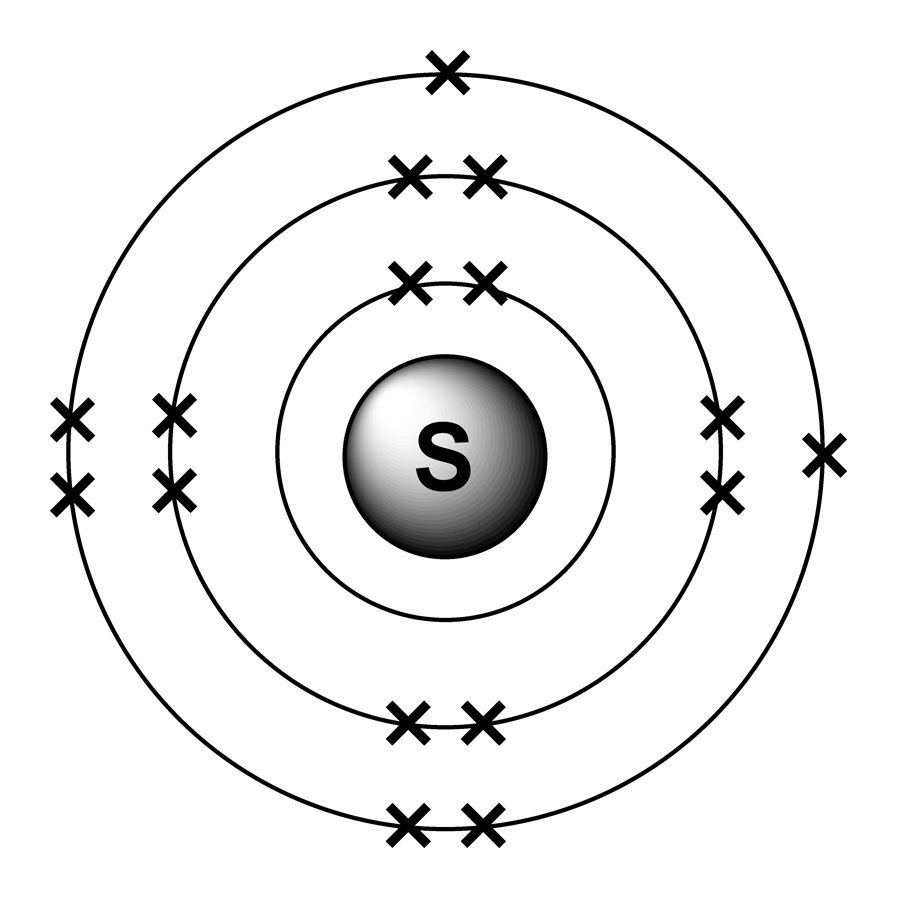
What is the electron configuration of silicon?
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.
Konsep Populer 19+ 1 Atome De Silicium
Silicon (Si) atom electron configuration (Bohr model) Electron configuration through orbitals follows different principles. For example Aufbau principle, Hund's principle, and Pauli's exclusion principle. Silicon electron configuration through orbit Scientist Niels Bohr was the first to give an idea of the atom's orbit.
FileThis shows the bond of Silicon Oxide using the Bohr model.png
Simplified Bohr model of a silicon atom, with one valence electron excited to the 1st excited energy level. As the diagram shows, the excited electron leaves behind an 'electron-hole' in the valence energy level.Through various mechanisms, this excited electron will lose energy, fall back to the valence energy level, and recombine with its electron-hole.
silicon atomic model 3d model of silicon atom QFB66
Silicon is a semiconductor and copper is a conductor. Bohr diagrams of the silicon atom and the copper atom are shown in Fig. 4. Figure 4: Bohr diagrams of the silicon and copper atoms. The core of the silicon atom has a net charge of 4 (14 p +, 10 e-) and the core of the copper atom has a net charge of 1 (29 p +, 28 e-). The core includes.

Image result for silicon atomic model
Here, we will draw the Bohr diagram of the Silicon atom with some simple steps. Steps to draw the Bohr Model of Silicon atom 1. Find the number of protons, electrons, and neutrons in the Silicon atom Protons are the positively charged particles and neutrons are the uncharged particles, both these are constituents of the atom nuclei.
Silicon Facts St. Charles
1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).